Home

Elizabeth A. Ronan, PhD
Postdoctoral Researcher
University of Michigan School of Dentistry
Welcome! I am a neuroscientist studying the molecular and cellular mechanisms of orofacial somatosensation and pain. My research integrates insights from both invertebrate and vertebrate model systems to understand how sensory neurons detect, encode, and transmit information that drives perception, behavior, and homeostasis.
Recent Highlights
| Mar 10, 2026 | Publication — Rodents love to gnaw… which can be cute or annoying (e.g., when they decided to destroy my car wiring). But our new collaborative study published in Neuron uncovers why. Led by Dr. Xinyu Su and Bo Duan Lab, we discovered that incisor tactile input is relayed by a sensorimotor-motivational circuit to drive gnawing. Congratulations to all the authors! Featured in UMich News and Science. |
|---|---|
| Mar 01, 2026 | Publication — Our protocol for precise activation of intradental neurons in mice via single-molar electrical stimulation is now published in STAR Protocols! We developed a precision electrical stimulation method to overcome the technical barriers to targeting sensory innervation within the mineralized tooth, providing a robust tool for studying tooth sensation and pain using model organisms. |
Selected Publications
-
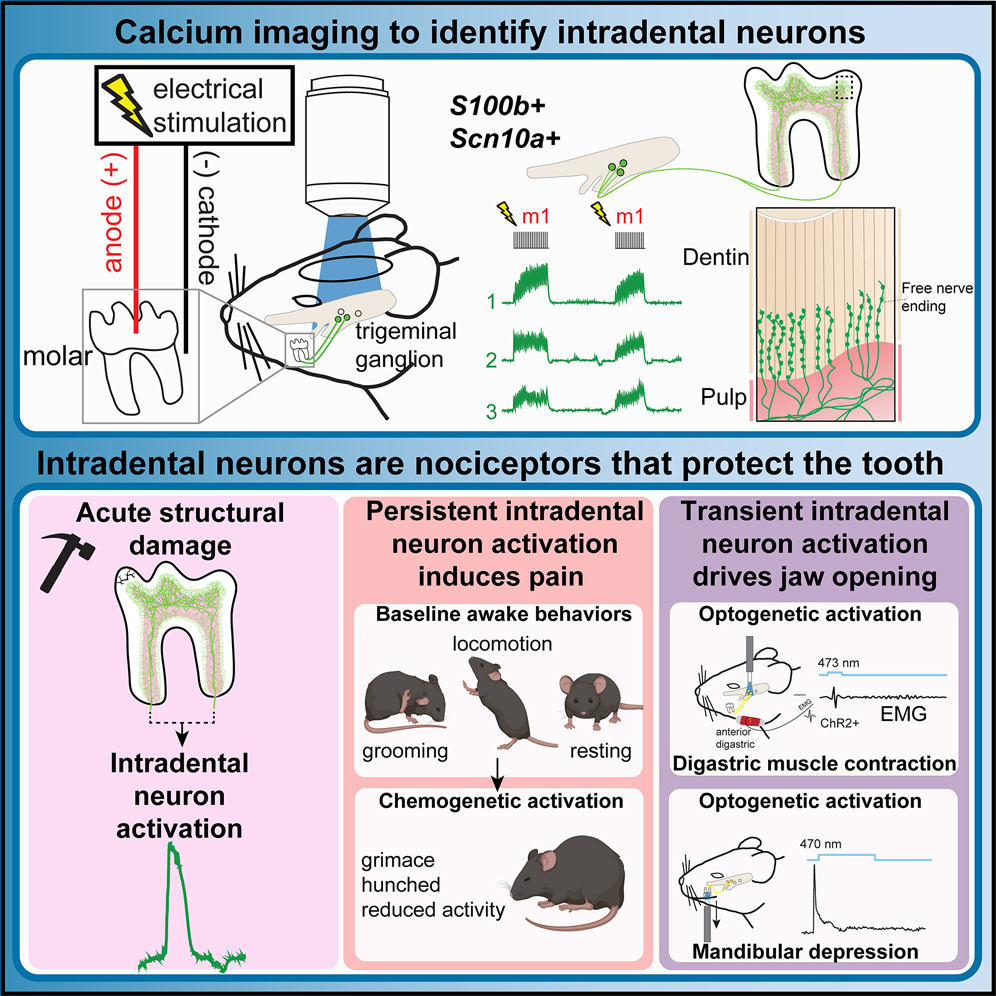 Intradental mechano-nociceptors serve as sentinels that prevent tooth damage.Cell Reports, Aug 2025
Intradental mechano-nociceptors serve as sentinels that prevent tooth damage.Cell Reports, Aug 2025EXPAND ABSTRACT
Abstract: The trigeminal sensory neurons that innervate the tooth’s vital interior—intradental neurons—are expected to drive severe pain, yet their contribution to healthy tooth sensation has not been explored. Here, we uncover a role for myelinated high-threshold mechano-nociceptors (intradental HTMRs) in tooth protection using in vivo Ca2+ imaging, opto-/chemogenetics, and the AI-driven behavioral analysis tool LabGym. Intradental HTMRs innervate the inner dentin through overlapping receptive fields and respond as the external structures of the tooth are damaged in the absence of either PIEZO2 or Nav1.8. Whereas chemogenetic activation of intradental HTMRs results in a pain phenotype marked by facial and postural changes, their transient optogenetic activation triggers a rapid, jaw-opening reflex via contraction of the digastric muscle. Our work indicates that intradental HTMRs not only trigger pain but also protect the teeth by initiating a reflexive movement of the jaws when the teeth experience damage during chewing. -
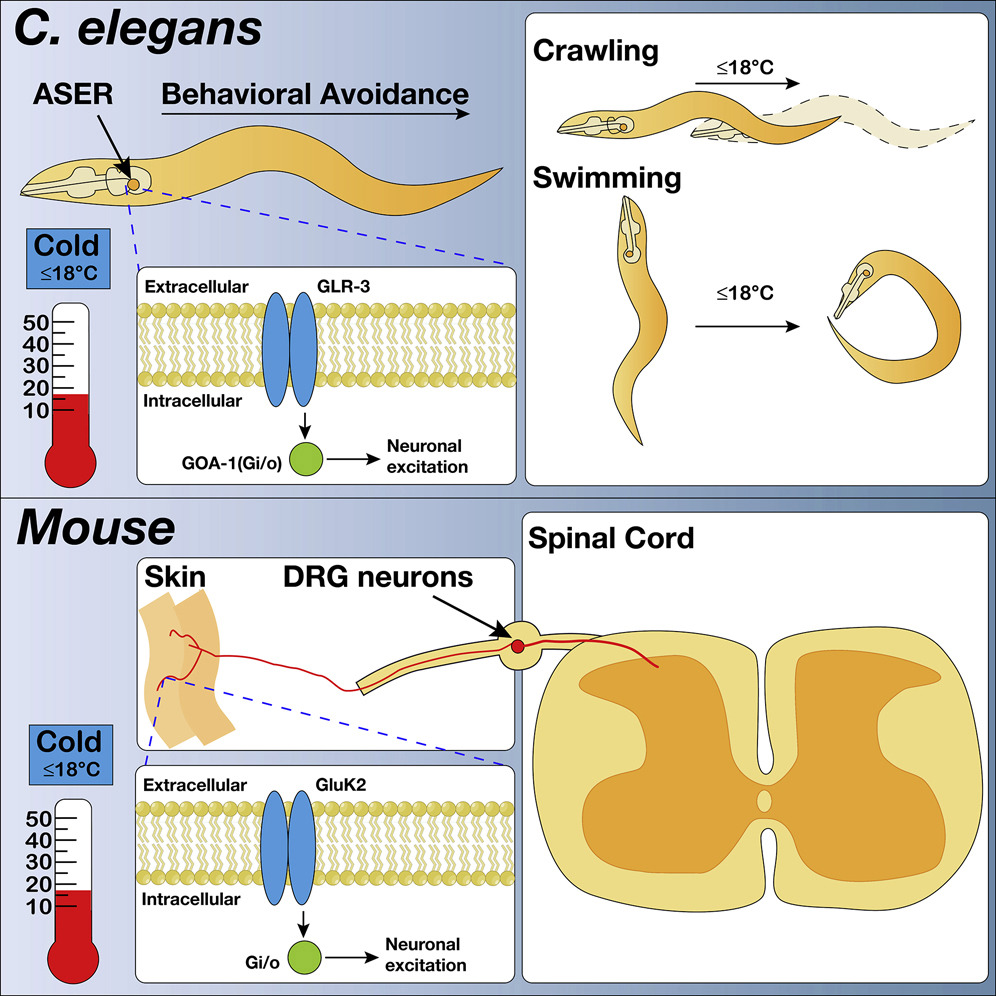 A Cold-Sensing Receptor Encoded by a Glutamate Receptor Gene.Cell, Sep 2019
A Cold-Sensing Receptor Encoded by a Glutamate Receptor Gene.Cell, Sep 2019EXPAND ABSTRACT
Abstract: In search of the molecular identities of cold-sensing receptors, we carried out an unbiased genetic screen for cold-sensing mutants in C. elegans and isolated a mutant allele of glr-3 gene that encodes a kainate-type glutamate receptor. While glutamate receptors are best known to transmit chemical synaptic signals in the CNS, we show that GLR-3 senses cold in the peripheral sensory neuron ASER to trigger cold-avoidance behavior. GLR-3 transmits cold signals via G protein signaling independently of its glutamate-gated channel function, suggesting GLR-3 as a metabotropic cold receptor. The vertebrate GLR-3 homolog GluK2 from zebrafish, mouse, and human can all function as a cold receptor in heterologous systems. Mouse DRG sensory neurons express GluK2, and GluK2 knockdown in these neurons suppresses their sensitivity to cold but not cool temperatures. Our study identifies an evolutionarily conserved cold receptor, revealing that a central chemical receptor unexpectedly functions as a thermal receptor in the periphery. -
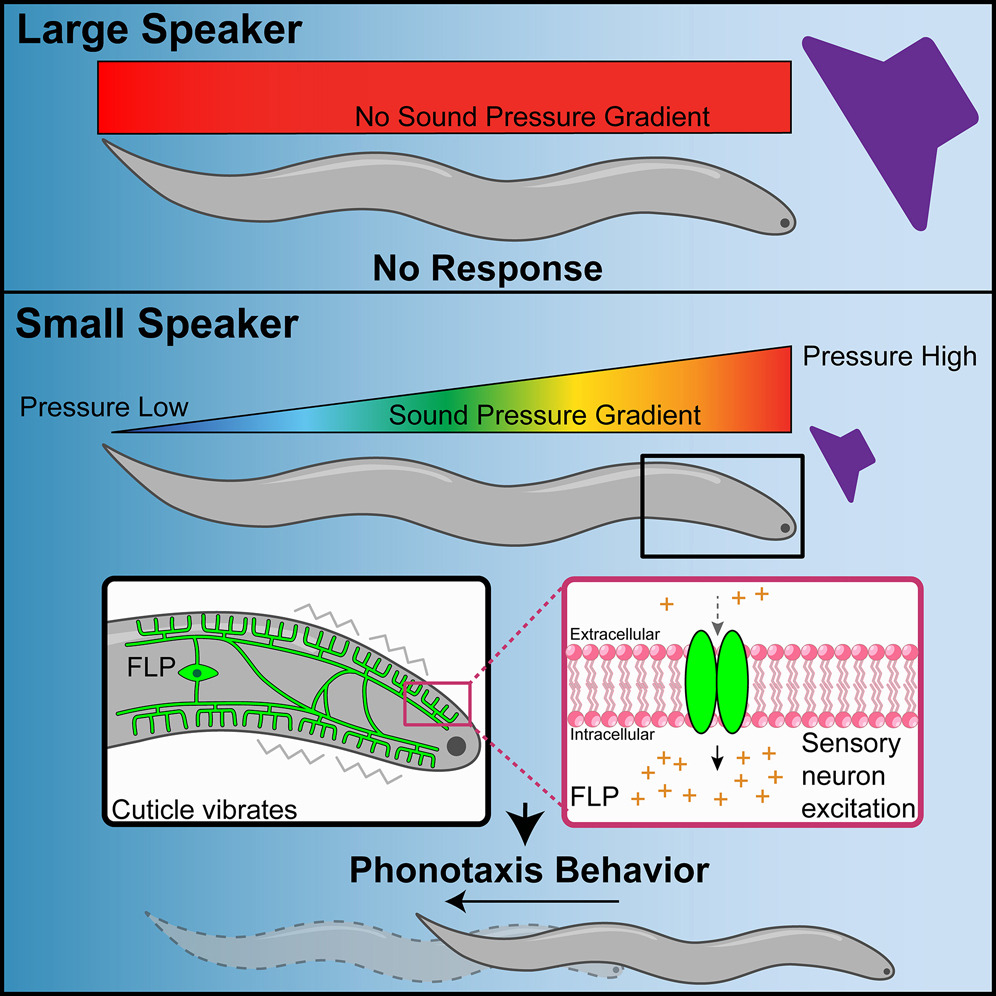 Sensing of sound pressure gradients by C. elegans drives phonotaxis behavior.Current Biology, Sep 2023
Sensing of sound pressure gradients by C. elegans drives phonotaxis behavior.Current Biology, Sep 2023EXPAND ABSTRACT
Abstract: Despite lacking ears, the nematode C. elegans senses airborne sound and engages in phonotaxis behavior, enabling it to locate and avoid sound sources. How worms sense sound, however, is not well understood. Here, we report an interesting observation that worms respond only to sounds emitted by small but not large speakers, indicating that they preferentially respond to localized sound sources. Notably, sounds emitted by small speakers form a sharp sound pressure gradient across the worm body, while sounds from large speakers do not, suggesting that worms sense sound pressure gradients rather than absolute sound pressure. Analysis of phonotaxis behavior, sound-evoked skin vibration, and sound-sensitive neuron activities further support this model. We suggest that the ability to sense sound pressure gradients provides a potential mechanism for worms to distinguish sounds generated by their predators, which are typically small animals, from those produced by large animals or background noise. As vertebrate cochlea and some insect ears can also detect sound pressure gradients, our results reveal that sensing of sound pressure gradients may represent a common mechanism in auditory sensation across animal phyla.
Latest Posts
No posts so far...